![PDF] Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) checklist | Semantic Scholar PDF] Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) checklist | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/05474966fa1f0196292dace9ec79649a351cb2d7/5-Figure4-1.png)
PDF] Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) checklist | Semantic Scholar
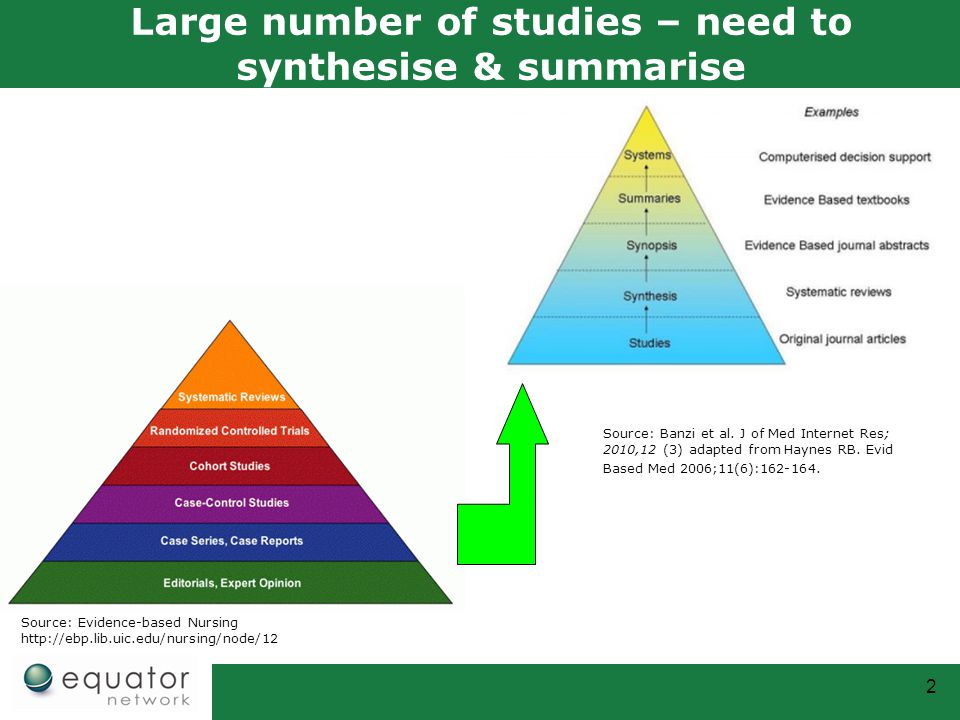
IJERPH | Free Full-Text | Mental Health of Children and Adolescents Amidst COVID-19 and Past Pandemics: A Rapid Systematic Review
Reporting Guidelines and the American Journal of Public Health's Adoption of Preferred Reporting Items for Systematic Reviews
![PDF] Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement | Semantic Scholar PDF] Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/245831b1ba9fa32fdb224555b37533010af903e6/4-Table2-1.png)
PDF] Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement | Semantic Scholar

Preferred reporting items for systematic reviews and meta-analyses: the... | Download Scientific Diagram

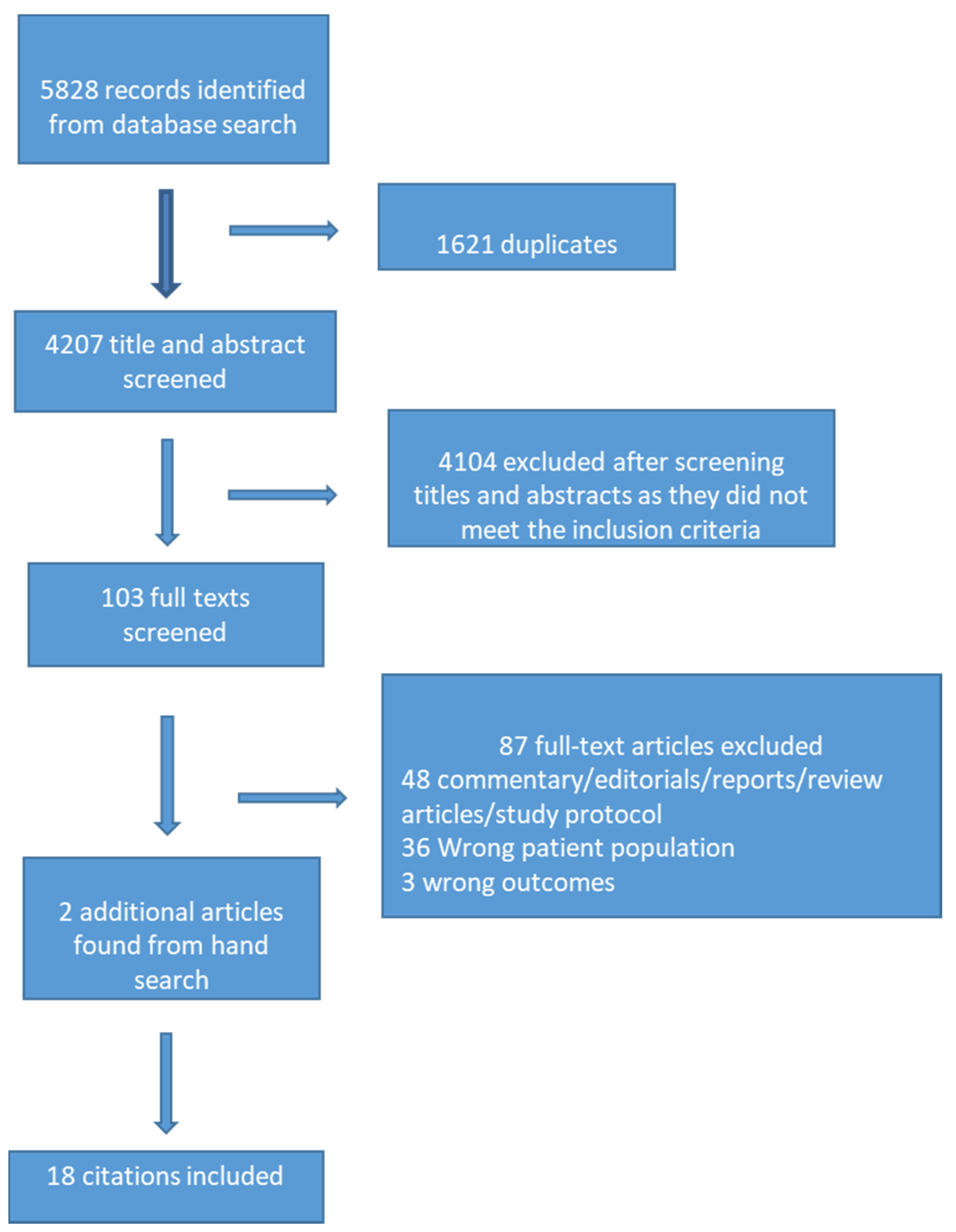
Individual Participant Data Meta-analysis - Everything you always wanted to know ... | Cochrane Methods
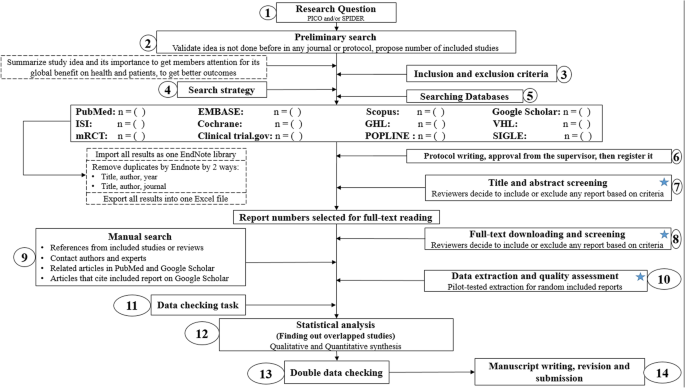
A step by step guide for conducting a systematic review and meta-analysis with simulation data | Tropical Medicine and Health | Full Text
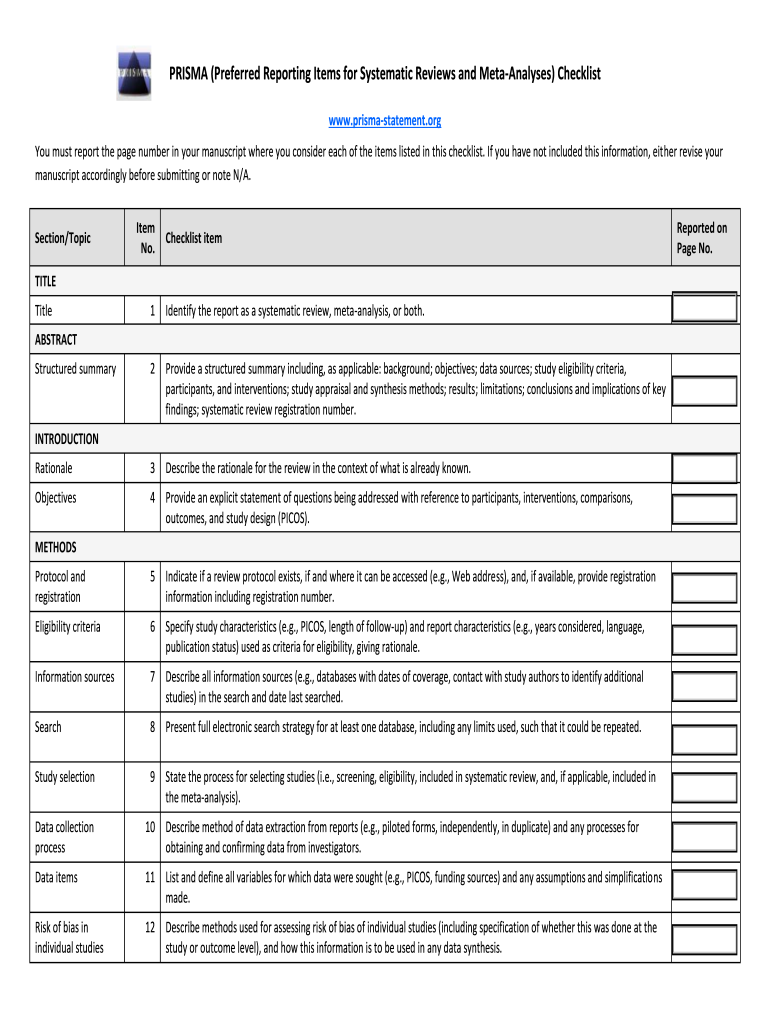
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) Checklist: Fill out & sign online | DocHub
Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement | PLOS Medicine

The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration - ScienceDirect
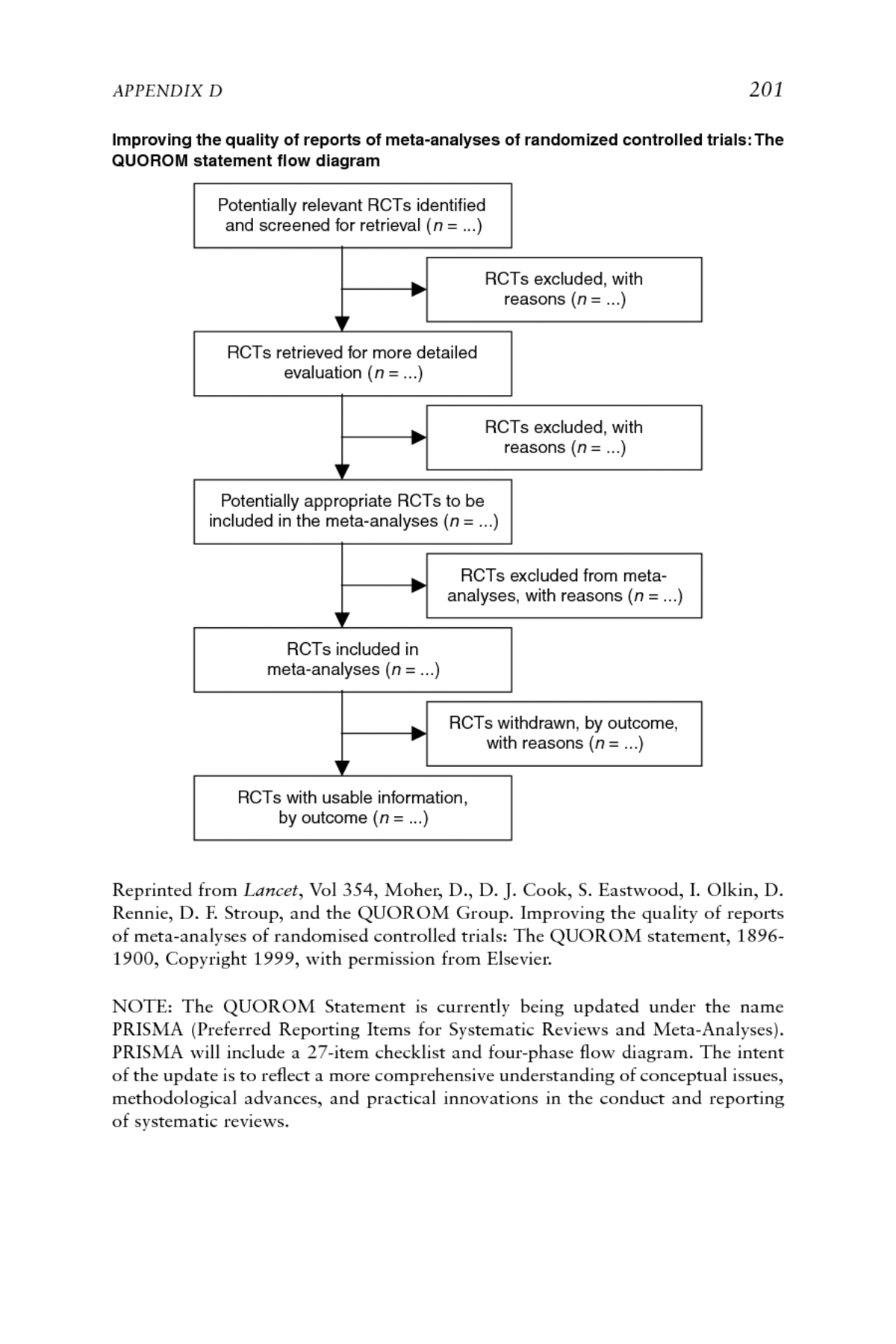
Appendix D: Standards for Reporting Meta-Analyses of Clinical Trials and Observational Studies: QUOROM and MOOSE | Knowing What Works in Health Care: A Roadmap for the Nation |The National Academies Press

Figure 1 from Preferred reporting items for systematic reviews and meta- analyses: the PRISMA Statement | Semantic Scholar
![PDF] Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement | Semantic Scholar PDF] Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/245831b1ba9fa32fdb224555b37533010af903e6/3-Table1-1.png)